Sodium Hypochlorite Generator
Sodium hypochlorite generators produce low strength disinfectants on demand through the electrolysis of brine solutions. On-site hypochlorite generation systems use only salt, water and electricity to safely generate sodium hypochlorite on demand.
Designed to replace liquid chlorine delivery and chlorine tank storage in most water treatment applications, NaClO generators produce a strong oxidizing solution that kills Staphylococcus aureus and E. coli organisms as well as other harmful pathogens.
How does sodium hypochlorite generator work?
Sodium hypochlorite generators are used in pre-engineered on-site systems to safely and effectively produce chlorine. Converts a saturated brine solution into hypochlorite at a concentration of 0.8% – below the 1% hazardous substance threshold. The system includes a brine tank, chlorine storage tank and control unit.

The sodium hypochlorite generator alleviates the safety concerns associated with the storage and use of bulk sodium hypochlorite, chlorine tablets or chlorine gas. By generating chlorine on site, the generator produces sodium hypochlorite at a significantly lower cost than purchasing hypochlorite externally, and hypochlorite stays fresh rather than degrading in storage. Ideal applications range from drinking water disinfection and the agri-food and beverage industry to other commercial industrial applications.
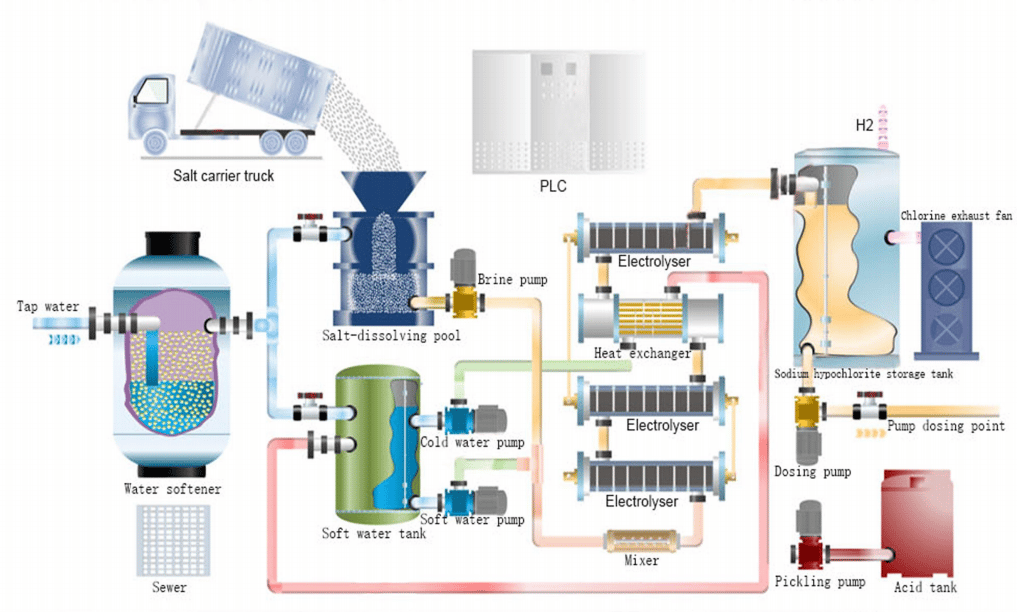
The process flow of conventional hypochlorite generator includes the following steps: First, the tap water is softened to remove calcium and magnesium ions from the water, and part of the softened water provides dilution water for the sodium salt feed. Hypochlorite generator; secondly, another part flows into the dissolved salt tank, which dissolves the refined salt into 30% saturated brine, and after softening, it is precisely mixed with diluted water through a metering pump, and then enters the power plant for DC electrolysis under the action of the electrolytic cell. The prototype in the cell produces chlorine gas and the cathode produces sodium hydroxide and hydrogen gas. The two products react to form sodium hypochlorite solution. The reaction equations are as follows.
Anodic reaction: 2Cl- – 2e- → Cl2 (Oxidation reaction)
Cathodic reaction: 2Na+ + 2e + 2H2O → 2NaOH + H2↑ (Reduction reaction)
Total reaction equation: NaCl+H2O → NaClO+H2↑
Because the molecular weight of Naclo is 1.05 times that of Cl,and the number of charge transfer between each NaClO molecule and Cl2, is the same when the oxidation reaction occurs. Therefore, in the process of producing sodium hypooxide through the electrochemical reaction rate path,every 1g of NaCIO produced is equivalent to 0.953g of available chlorine.

KUOSI is a manufacturer and supplier of on site sodium hypochlorite generator in China. Feel free to contact us for a quote on sodium hypochlorite generator systems.
Technical indicators
- Dilute brine concentration: 3%or directly use seawater
- Consumption of available chlorine salt per 1kg: <3.2Kg
- Power consumption per 1kg of available chlorine:<4.0kwh.
- Available chlorine concentration in the preparation solution: 6000-9000ppm(0.6%-0.9%), current efficiency: >78%
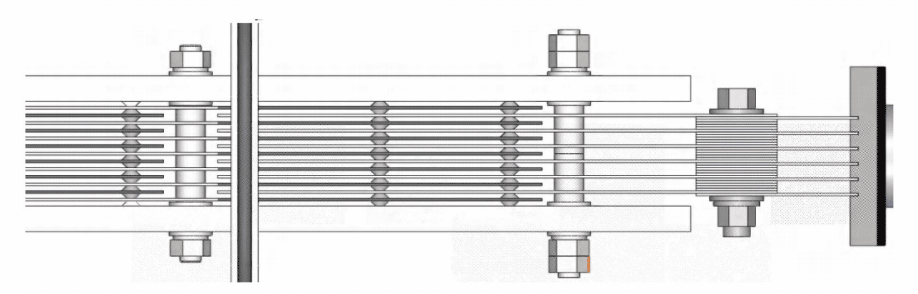
- Electrode materials: pure titanium materials(cathode, anode, fasteners), anode protective coating: mixed formula of rare and precious metal oxides such as ruthenium and iridium
- Disinfectant sterilization rate: 99.9%
- Service life of the electrolytic cell: The electrolytic cell is made of high anti-corrosion PMMA plexiglass and has a service life of more than 20 years
- Automatic control level: DSP console centralized control, large-scale system with remote monitoring and operation.
Features
- Intelligent algorithm prompts electrode life and acid cleaning cycle
- Access to the Internet of Things: remote intelligent identification and management, equipment failure warning and remote processing
- Intelligent control: automatic control, unattended, big data information collection
- Modular design: modular integrated equipment, fast and convenient equipment installation and maintenance
- High efficiency and low power consumption: low salt consumption, low power consumption, high current efficiency, high product liquid stability
- Safety and stability: corrosion-resistant materials + special coating, higher service life (normal service life of cathode > 20 years, normal service life of anode > 5 years), long-term stable operation
Applications
- Municipal waterworks
- Independent water suppliers
- Industrial wastewater plants and process water
- Cooling towers
- Swimming pools
- Water reuse
- Food processing
- Petrochemicals
- Power and energy
- Hospital wastewater

























